Super-Resolution Microscopy
Stochastic Optical Reconstruction Microscopy (STORM)
Photoactivated Localization Microscopy (PALM)
2014 Nobel Prize in Chemistry
Nature Methods: Method of the Year 2008
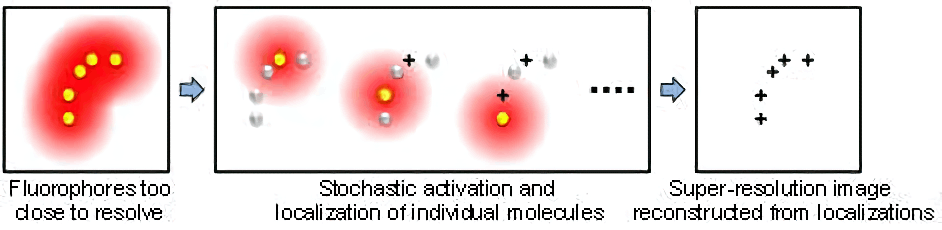
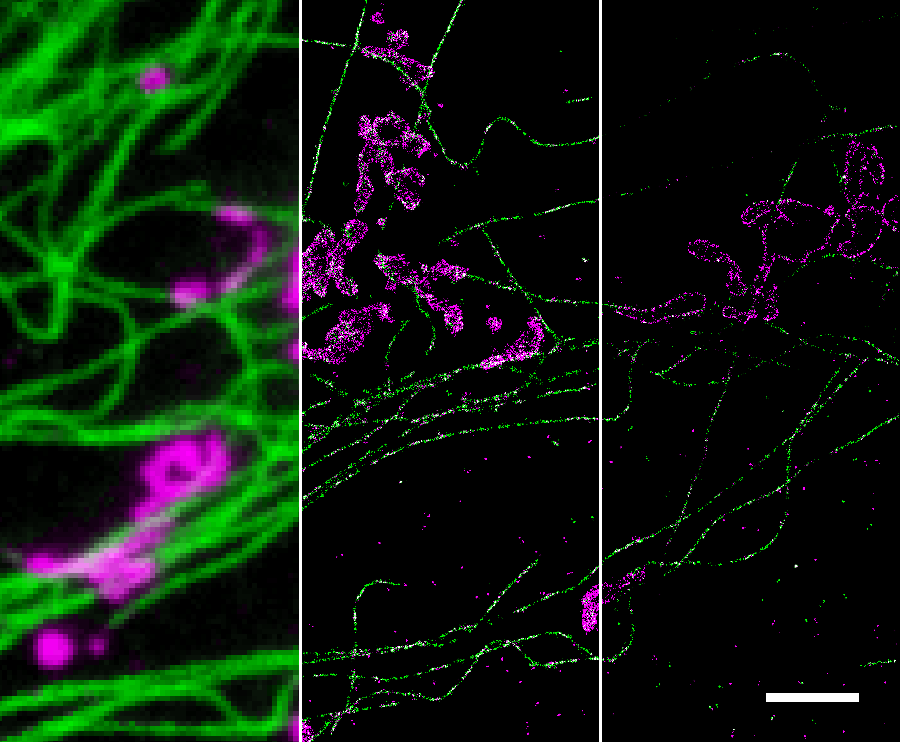
STORM (also named PALM) is a type of super-resolution optical microscopy technique based on stochastic switching of single-molecule fluorescence signal. In conventional fluorescence microscopy where all fluorophores in the sample are fluorescent, their diffraction limited images overlap, creating a smooth but blurred picture. STORM/PALM utilizes fluorescent probes that can switch between fluorescent and dark states so that in every snapshot, only a small, optically resolvable fraction of the fluorophores is detected. This enables determining their positions with high precision from the center positions of the fluorescent spots. With multiple snapshots of the sample, each capturing a random subset of the fluorophores, a final super-resolution image can be reconstructed from the accumulated positions. The scheme of STORM/PALM is illustrated below.